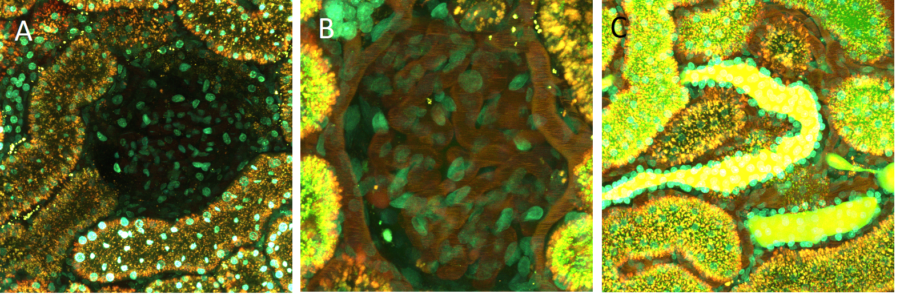
Still photomicrographs show the distribution of a large 150 Da-TRITC-dextran and a 4 kDa FITC-dextran at the surface of a living kidney. Panel A show an image taken prior to infusion of the dextrans. A glomerulus is seen in the center, surrounded by proximal tubules. The nuclei of all cells are seen in cyan. Panel B shows a 2-fold zoomed image of a glomerulus. The 150 kDa-TRITC-dextran is maintained in the circulating plasma which highlights the capillary loops of the glomerulus as well as the rapidly moving red blood cells (RBCs), which appear as dark streaks due to their rapid velocity. Panel C vividly shows two distal tubules (center) with the small 4k Da FITC-dextran filling the lumen of those tubules. In stark contrast, the lumens of the surrounding proximal tubules are largely devoid of the small dextran, instead showing accumulation of the 4k Da FITC-dextran in the early endosomes. Due to the FITC’s decreased fluorescence at low pH’s, there is little to no fluorescence emanating from late endosomes and lysosomes.
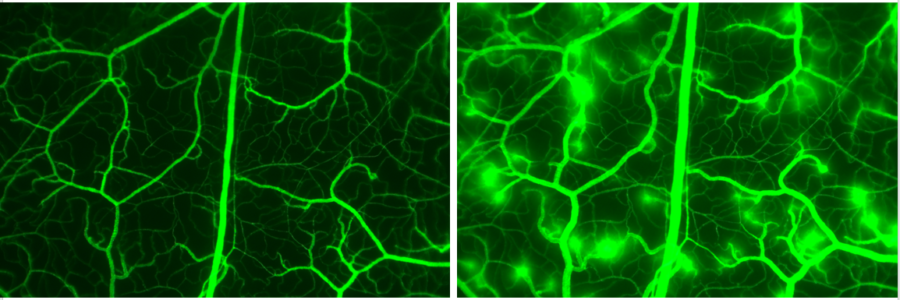
Histamine induced macromolecular (FITC-dextran) leakage in hamster cheek pouch. To the left: Control (1 min after FITC-dextran injection). To the right: Histamine infused leakage of 150 kDa FITC-dextran.




