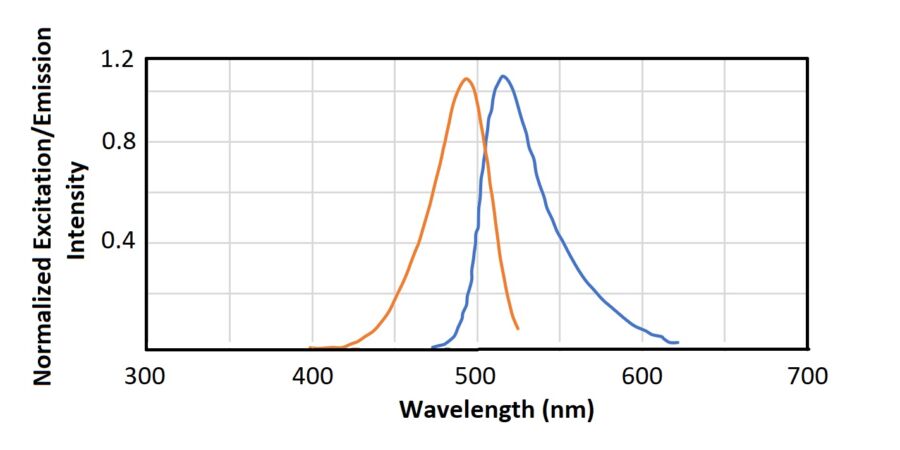
FITC and other fluorescent dyes have the ability of changing color in response to pH-changes. This can be utilized for measuring pH in living cells. Changes in cellular pH can reflect a range of physiological processes, including muscle contraction, endocytosis, cell proliferation, apoptosis, and ion transport11.
Fluorescent pH-indicators can be either stand-alone-dyes, such as FITC, or dyes coupled to a macromolecule, such as FITC-dextran. The advantage of using fluorescent dextran derivatives is that the molecules can be accumulated in specific intracellular compartments12. Compared to microelectrode techniques, fluorescent pH-indicators also have greater spatial sampling capability11. Another advantage of the probes and indicators is that they don’t bind to cellular proteins12.
FITC-products from TdB Labs
- FITC (isomer I)
- FITC -dextran
- FITC -lysine-dextran
- FITC-CM -dextran
- FITC-CM -polysucrose
- FITC-DEAE -dextran
- FITC-DEAE -polysucrose
- FITC -dextran sulfate
- FITC -hydroxyethyl starch
- FITC -inulin
- FITC -polysucrose
- FITC -Q-dextran
- FITC -trehalose
- Fluorescein Hyaluronic Acid
Read more about all applications here.